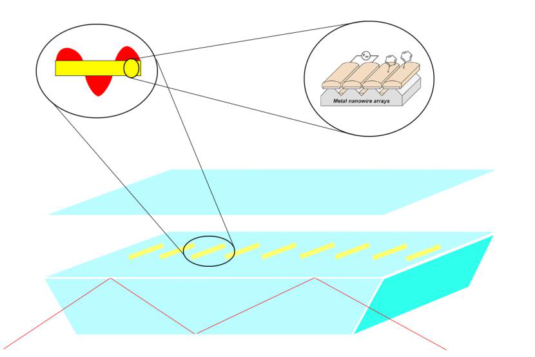
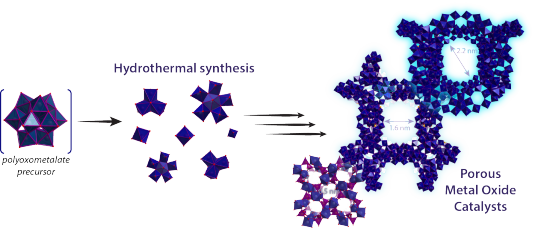
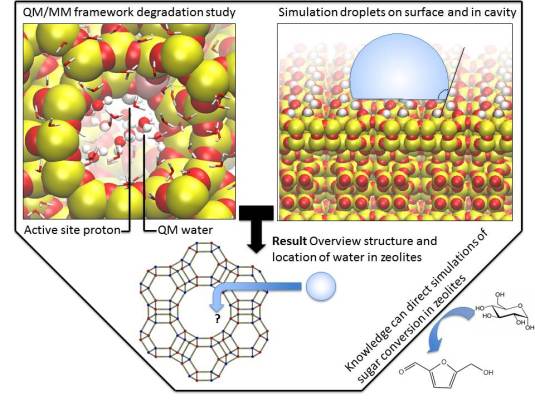
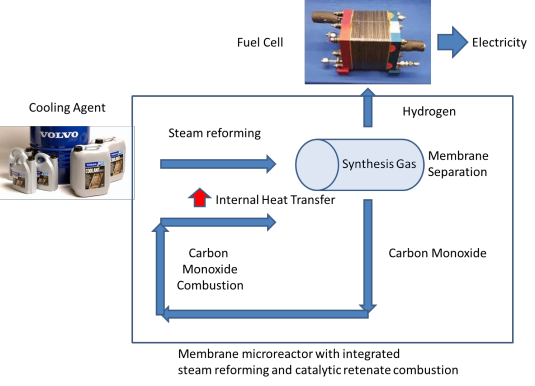
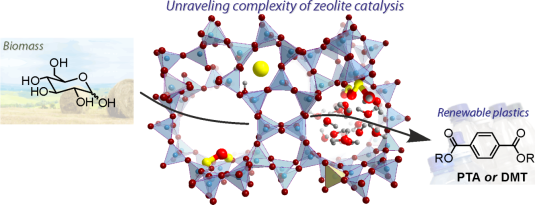
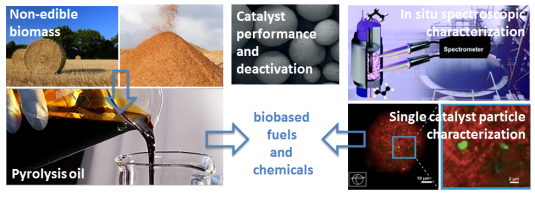
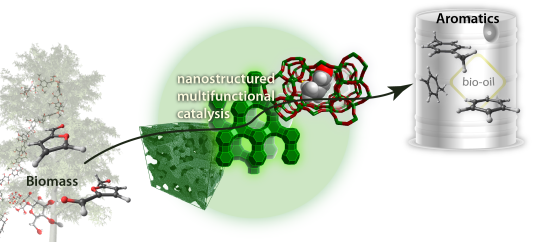
One of the issues in biomass conversion is the complexity of the mixtures involved. It is therefore difficult to do (direct) analysis of this mixture. Traditional methods like gas chromatography are not always suitable because of the high water content present within biomass. In this project, the aim is to develop a microfluidic chip for on-line analysis of the products formed during biomass conversion. This microfluidic chip will contain ordered nanofabricated structures for surface enhanced Raman spectroscopy (SERS), combined with additional structures for surface enhanced infrared absorption spectroscopy (SEIRAS). SEIRAS and SERS are two complementary methods, since vibrations active in SERS are inactive in SEIRAS and vice versa. Moreover, enhancement of the spectroscopic methods is essential to lower the limit of detection.
Project leader: Dr. Mathieu Odijk