Producing hydrogen from renewable energy sources such as solar energy is the missing link in the process of long-term energy storage and environment-friendly hydrogen vehicles. Heterogeneous catalysis can make this possible.
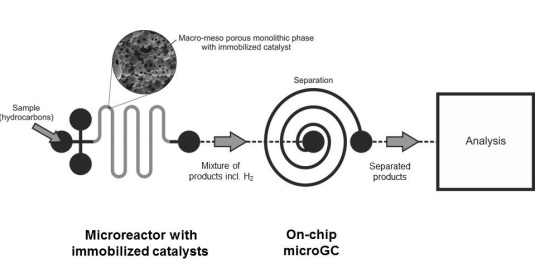
Heterogeneous catalysis is an interfacial phenomena, and consequently requires a high surface area to be efficient. Nanocatalysts have a very high surface/volume ratio due to their size, which means a high activity with a low amount of material. However once they are dispersed in a large reactor, controlling and recovering these nanocatalysts is extremely challenging. Nanocatalysts are usually embedded into porous materials that do not interfere with the chemical reactions. This matrix must have small pores to offer a large contact area between the nanocatalysts and the media, and pores large enough to enable a good transport of the reactants and the products. These two conditions can be fulfilled if the matrix presents a hierarchy of pore sizes.
We synthesize porous microbeads (100 µm) loaded with nanocatalysts (20 nm) and which presents different sizes of pore (about 1 µm). We use microfluidics to have a good control of the physical conditions and therefore of the pore formation. In our microfluidic chip, we create nanocatalysts-loaded droplets that are cooled down along their trajectory in the channels. When temperature drops, the components of the droplets demix and we use UV light to solidify one of the two phases of the droplets. When the second phase is rinsed we obtain porous microbeads. The alignment of the temperature gradient with the UV beam is our lever to tune the properties of our system of pores. We will then measure the catalytic activity of our beads and optimize it by adjusting the size of the pores that depends on the alignment of the UV beam.

Left: Three steps of microbeads production: initial homogeneous and hot droplet laoded with nanocatalysts, cold and demixing droplet, porous microbeads after solidification of the red phase and rinsing of the blue phase. Right: Scanning-electron-microscopy image of our material loaded with nanoparticles prepared in a vial (scale bar 2 µm).
Our first target application is ethanol reforming to produce hydrogen under illumination with sun or UV light. Several nanocatalyst are known to make this reaction possible such as platinum-titania nanoparticles. However our aim is to produce a versatile matrix so we will later use these microbeads with other kind of nanocatalysts to prove their versatility.
Project leader: Prof. Albert van den Berg